- About
-
Solutions
-
Services
- Biosciences
- Chemistry
- Integrated Drug Discovery
- Computer Aided Drug Design
- Hit ID and Screening Services
- DMPK Services
- Target Classes and Modalities
- Therapeutic Areas
-
A-Z
- A
- B
- C
- D
- E
- F
- G
- H
- I
- K
- L
- M
- N
- O
- P
- R
- S
- T
- V
- X
-
Services
- Library
- News & Events
- Careers
Biotherapeutics
deep expertise • flexible solutions • close collaboration • rapid delivery
Accelerate Your Biologics Discovery Program with Domainex
- Integrated biologics discovery support designed to accelerate and strengthen the development of modern biotherapeutics
- Highly collaborative approach with transparent communication from project initiation onward
- Rapid delivery of high‑quality reagents and data to support confident, well‑informed decision‑making
- Deep scientific expertise spanning protein production, analytics, structural biology and cellular characterisation
- Flexible workflows adapted to the needs of antibody, peptide, therapeutic protein, or other biologics modalities
- Deep scientific expertise spanning protein production, analytics, structural biology and cellular characterisation
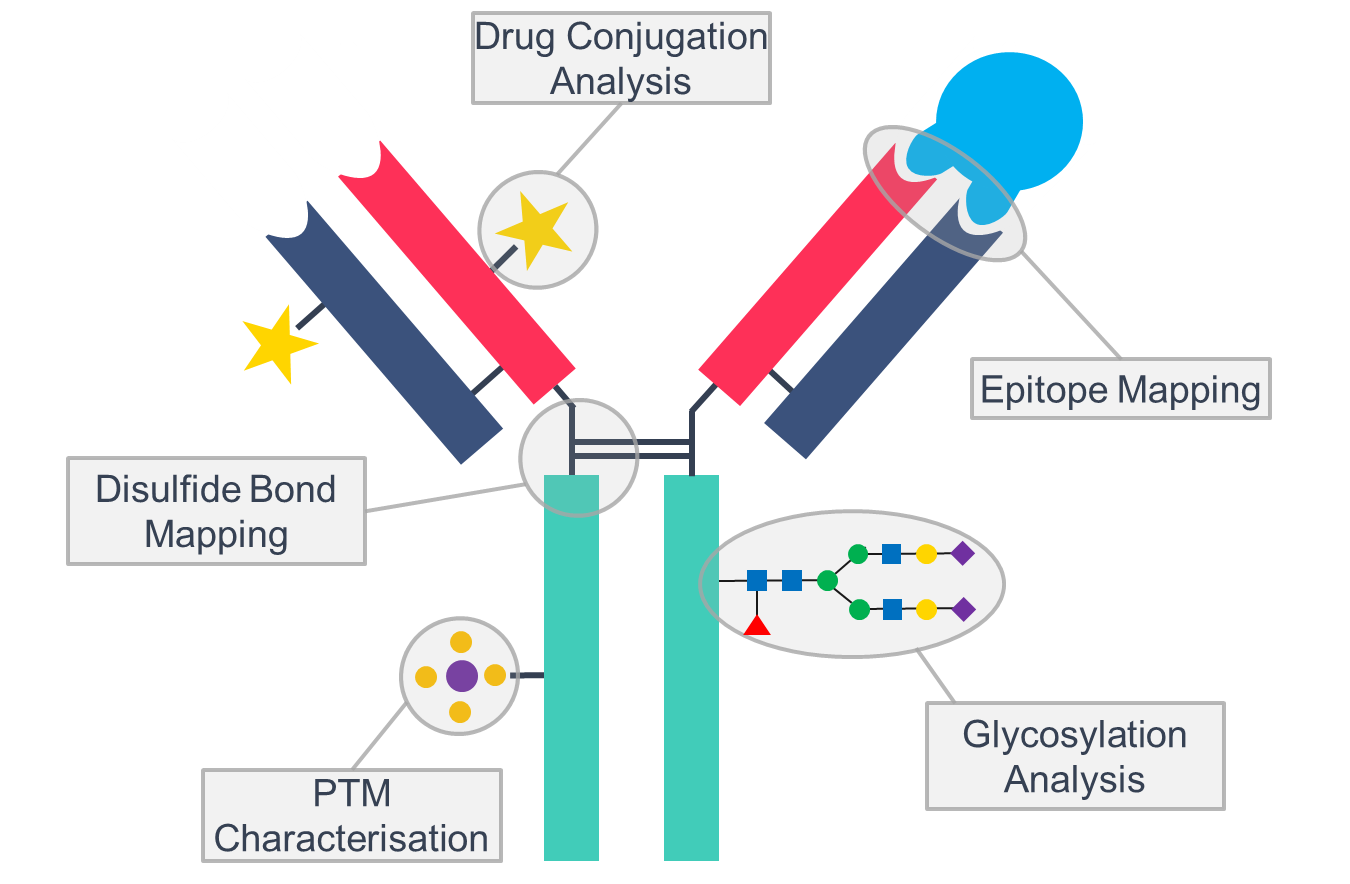
Figure 1. Schematic of an antibody with potential changes and characterisation
Protein Design & Production
- Collaborative construct design based on structural, sequence‑based and computational insights, including AI‑assisted approaches
- Production of soluble, membrane‑associated, and complex multidomain protein targets
- High‑quality reagents produced to meet stringent scientific and downstream application requirements
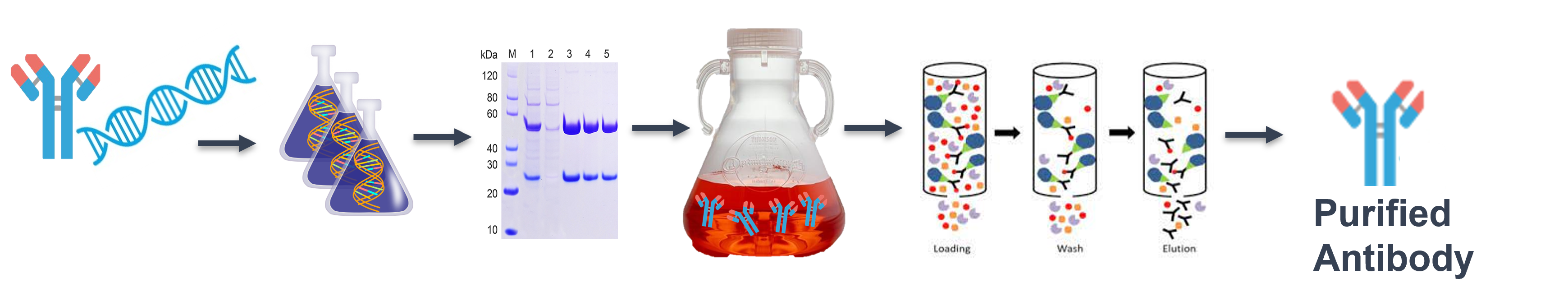
Figure 2. Antibody production workflow
Mammalian Expression Systems
- High‑quality antigen production in mammalian systems ensures native‑like folding, correct quaternary structure, and physiologically relevant glycosylation patterns including our membrane protein expression technology - essential for antibody recognition.
- Our antigen expression strategies preserve correct folding and essential PTMs, enabling accurate epitope presentation and increasing the success rate of downstream hit generation and screening
- Early expression screening in mammalian systems identifies scalable yields suitable for long‑term development
AI guided de-novo Biotherapeutics Design
Towards Universal Binder Design
- All-atom generative AI/ML modelling enables the design of proteins/peptides across all modalities
- State-of-the-art co-folding algorithms – validated by wet lab experiments
- Powered by our HPC and GPU based infrastructure

Figure 3. Rheumatoid Factor (RF) antibody designed in a computational study
Protein QC & Analytical Characterisation
Early developability assessment
- Evaluation of aggregation, homogeneity, and biochemical behaviour to de-risk therapeutic development
- Comprehensive QC analytics providing detailed insight into thermal stability, aggregation, solubility, identity, purity and structural integrity
- Flexible application of SEC‑MALS, NanoDSF/DSF, DLS, mass spectrometry, SDS‑PAGE, CE‑SDS and HPLC methodologies
- Rapid generation of reliable analytical data to support critical project decisions (glycosylation analysis, epitope mapping, etc)
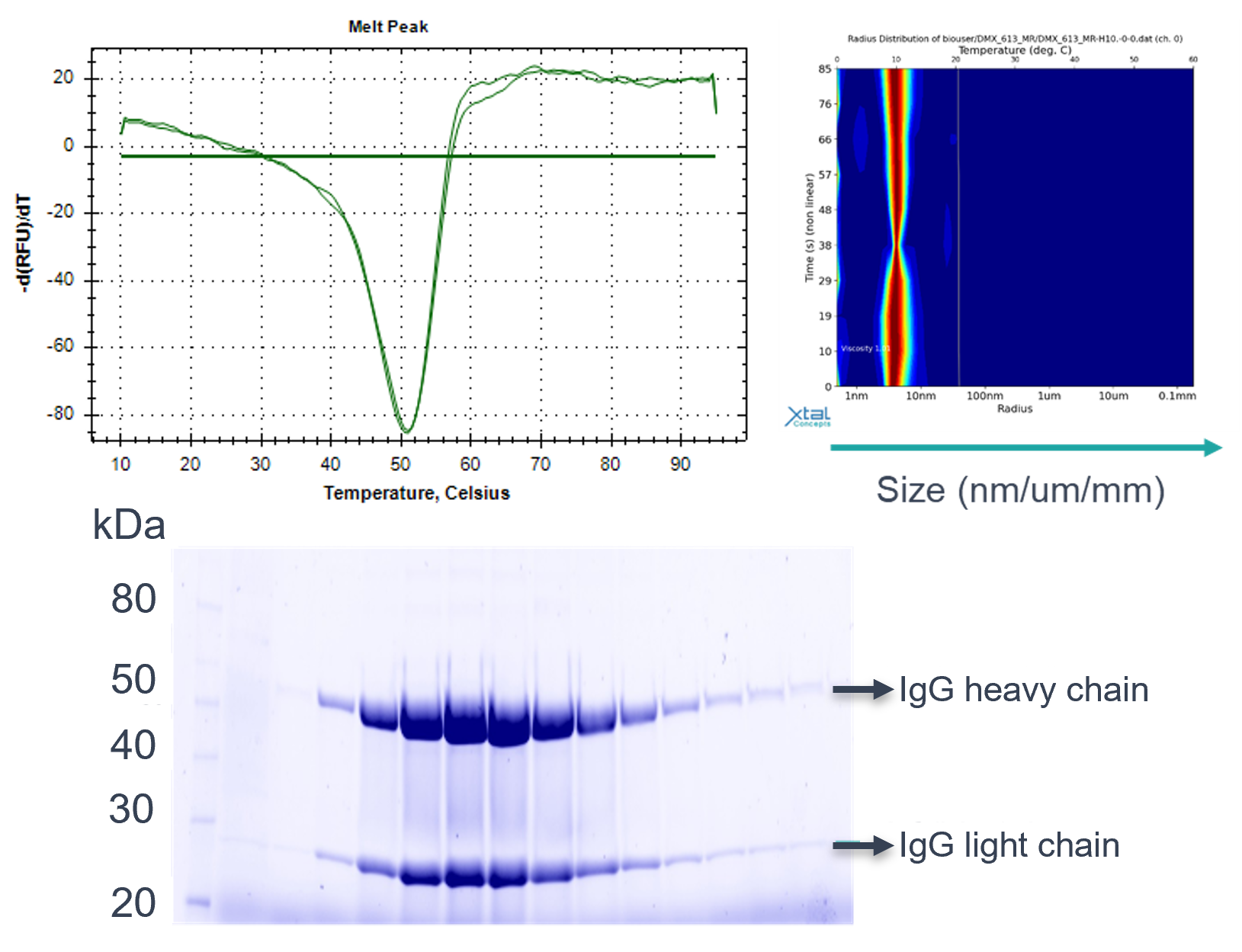
Figure 4. Examples of characterisation techniques, including SDS-PAGE, DSF and SEC
Biophysical Characterisation & Structural Biology
Collaborative assay strategy development tailored to each biologics programme
- A comprehensive suite of biophysical techniques, including GCI (SPR), BLI, spectral‑shift analysis, MST, ITC, native mass spectrometry and mass photometry
- Binding analysis from yes/no to ranking and detailed kinetic characterisation
- Epitope binning
- Binding specificity
- Protein QC, bioanalytical, binding kinetics and thermodynamic analysis to inform on early developability assessment
- Expert interpretation of complex biophysical and functional data for confident programme progression
Expertise across X‑ray crystallography, cryo‑EM and HDX-MS
- Rapid generation of high‑value structural insights for rational design and optimisation of biotherapeutics
- Flexible structural biology workflows tailored to proteins, peptides, antibodies and other modalities
- Detailed molecular understanding to guide engineering strategies and refine candidate selection
Cell‑Based Assays & Functional Characterisation
Collaborative design of biologically relevant cell‑based assays to assess function and mechanism
- Rapid evaluation of target engagement, pathway modulation and mechanism‑of‑action
- Flexible assay formats that align with programme goals and target biology, including primary cells where appropriate
- Expert analysis and interpretation to integrate molecular, structural and cellular findings
- Assessment of receptor internalisation to support modalities such as antibody–drug conjugates (ADCs), targeted payload delivery, or receptor modulation strategies
- Biomarker assay development and screening to support mode‑of‑action studies, pharmacodynamic assessment and translational strategies
Frequently Asked Questions
1. What are the timelines for antibody production?
The timeline for delivery of purified antibodies usually within 2-3 weeks but can vary depending on the nature of the target
2. What are the QC methods used?
QC methods for purified antibodies include SDS-PAGE, western blotting, size-exclusion chromatography (SEC), DLS, mass photometry, ELISA and immunoprecipitation
Start your next project with Domainex
Contact one of our experts today