- About
-
Solutions
-
Services
- Biosciences
- Chemistry
- Integrated Drug Discovery
- Computer Aided Drug Design
- Hit ID and Screening Services
- DMPK Services
- Target Classes and Modalities
- Therapeutic Areas
-
A-Z
- A
- B
- C
- D
- E
- F
- G
- H
- I
- K
- L
- M
- N
- O
- P
- R
- S
- T
- V
- X
-
Services
- Library
- News & Events
- Careers
Fibrosis Assays
deep expertise • flexible solutions • close collaboration • rapid delivery
Pioneering Cellular Models for Fibrosis, Inflammation, and Regenerative Biology
Fibrotic diseases are characterised by excessive extracellular matrix (ECM) deposition, persistent inflammation, and aberrant tissue remodelling leading to dysregulated wound healing. These mechanisms underpin a variety of debilitating conditions, including pulmonary fibrosis, fibrotic interstitial lung diseases, liver and cardiac fibrosis, and kidney fibrosis.
Key to this physiology is the interplay between immune cells, fibroblasts, and epithelial or endothelial cell types. Macrophages, T cells, dendritic cells, and natural killer (NK) cells modulate fibroblast activation and myofibroblast differentiation through signalling pathways including TGF‑β/Smad, NF‑κB, JAK/STAT, and PI3K/AKT. These reciprocal interactions regulate ECM turnover, tissue stiffness, inflammatory persistence, and repair quality across diverse organs.
To support client programmes we offer a number of physiologically relevant and scalable human in vitro models that replicate key mechanisms in fibrosis. These platforms enable our clients to accelerate the discovery, validation, and optimisation of next‑generation therapeutics.
Functional Fibrosis Readouts
Domainex have access to healthy and diseased primary human fibroblasts and epithelial cells and a wealth of experience in functional fibrosis readouts.
Scar in the Jar Assay
- Macromolecular crowding to accelerate models of extracellular matrix deposition
- Quantification of collagen I and III and fibronectin deposition
- Multiplexed readouts allow parallel monitoring of matrix deposition, cell proliferation and cytokine secretion
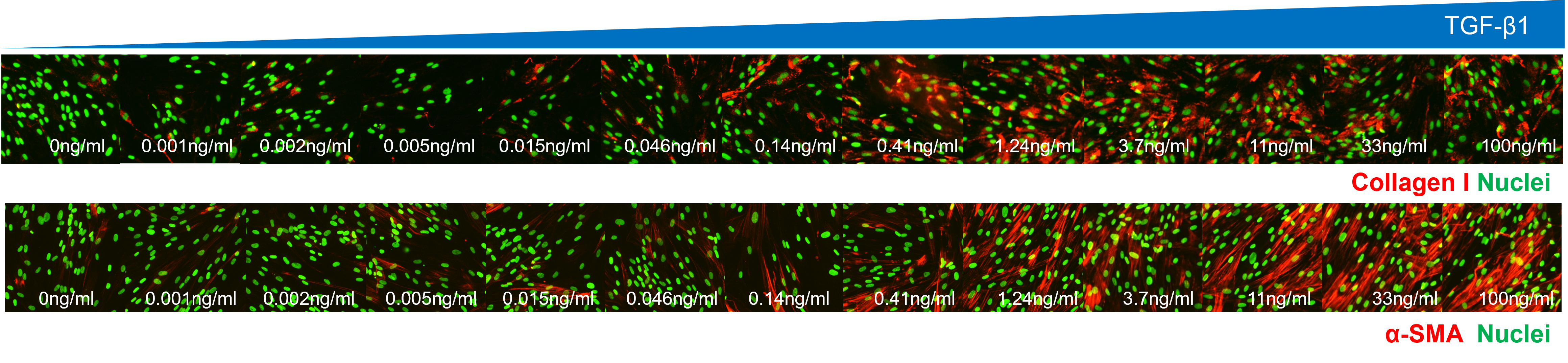
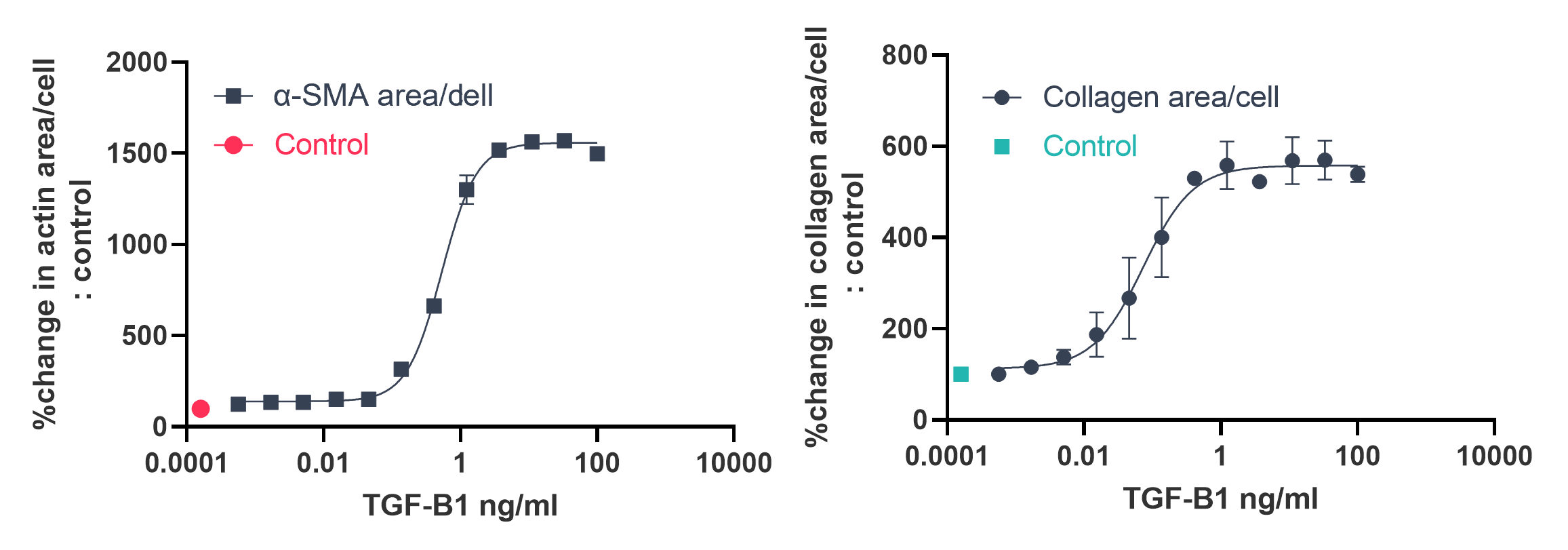
Example Data:
- Human fibroblasts treated with varying concentrations of TGF-β1 for 72 hours in Ficoll media (n=2)
- Immunostaining with either anti-collagen I or anti-α-SMA antibody
Fibroblast‑to‑Myofibroblast Transition (FMT) Assay
- Quantification of fibroblast differentiation into a pro-fibrotic, pro-proliferative myofibroblast phenotype
Epithelial‑to‑Mesenchymal Transition (EMT) Assay
- Quantification of matrix production from epithelial cells differentiating into a mesenchymal phenotype
Barrier Function Assays
- Using epithelial and endothelial systems, to provide barrier analytics:
- TEER‑based and fluorescent protein diffusion-based assessment of barrier integrity
- Tight‑junction and barrier protein quantification (e.g. ZO-1, filaggrin)
- Injury‑repair and inflammatory challenge models
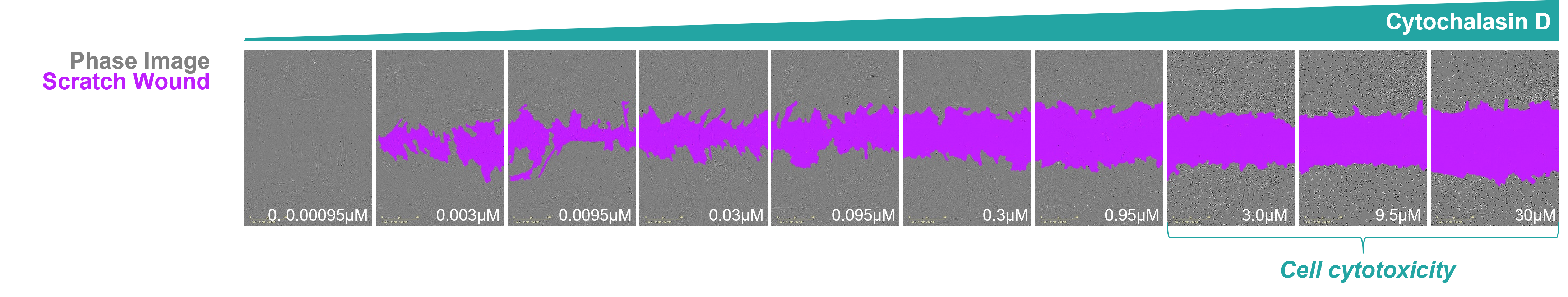
Scratch wound assay to measure epithelial or fibroblast wound healing
- Monitor uniform wound closure progression in real time using Sartorius Incucyte® S3
Pathway activation
Quantitative assessment of classic pro-fibrotic pathways such as phospho-SMAD2/3 pSMA2/3 at the protein or mRNA level.
Why Partner with Domainex?
Flexibility
Every assay at Domainex is custom‑designed: cell types, matrix components, cytokine environments, multi‑cellular co‑cultures can be adapted to answer your questions specific to the biological and therapeutic end point.
Deep Scientific Expertise
Domainex’s team combines decades of experience in fibrosis biology, immune‑stromal interactions, and advanced cellular modeling, aligned with current mechanistic insights from immunology and fibrotic cell research.
Collaborative Partnership
We function as an extension of your internal R&D team, ensuring transparency, rapid iteration, and scientifically rigorous data interpretation throughout every project stage.
Rapid Turnaround
Responsive rapid turnaround times and across our fibrosis model platforms enabling quicker decision-making and streamlined preclinical progression.
Start your next project with Domainex
Contact one of our experts today