
An integrated contract research organisation (CRO) accelerates drug discovery by providing end-to-end scientific expertise. From protein production and hit identification through lead optimisation and candidate selection – all under a single, coordinated and flexible partnership. This eliminates delays in data hand off, knowledge silos, and inefficiencies that arise when multiple specialist vendors are managed independently. For biotech companies, pharmaceutical organisations, and academic groups seeking to move from target to candidate faster and more cost-effectively, an integrated CRO model offers a proven strategic advantage.
What is an integrated drug discovery CRO?
An integrated drug discovery CRO is a contract research organisation that offers the full spectrum of pre-clinical drug discovery services within a single organisation. Rather than providing isolated capabilities such as medicinal chemistry or screening alone, an integrated CRO combines multidisciplinary expertise across biology, chemistry, structural biology, computational science, and DMPK - under one roof with a unified project team managing the programme from start to finish.
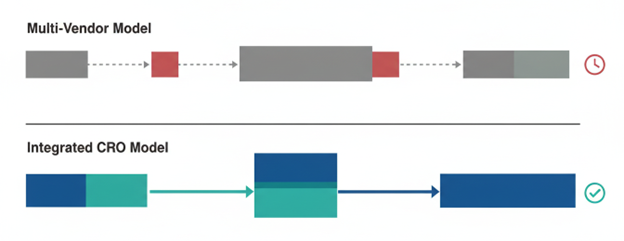
This model differs fundamentally from the traditional outsourcing approach, where a sponsor company contracts separate specialist CROs for each stage of discovery - one for protein production, another for assay development, a third for medicinal chemistry, and so on. While each individual vendor may deliver excellent work, the cumulative cost of managing multiple relationships, transferring data between organisations, and re-establishing project context at every handoff creates significant drag on timelines and budgets.
| Model | Coordination | Data Flow | Timeline Impact |
|---|---|---|---|
| Multi-vendor outsourcing | Sponsor manages multiple contracts and relationships | Data transferred between organisations at each stage; risk of information loss | Delays at every handoff; sponsor bears full project management burden |
| Integrated CRO partnership | Single dedicated project leader; one contractual relationship | Seamless internal data sharing across disciplines in real time | Faster decision-making; continuous momentum from hit ID to candidate selection |
What are the key benefits of using a CRO throughout drug discovery?
The benefits of partnering with an integrated CRO across the full drug discovery process fall into five core areas: speed, cost efficiency, scientific quality, risk reduction, and strategic flexibility.
1. Faster progression from target to candidate
When biology, chemistry, structural biology, and ADMET teams operate within the same organisation, structure-activity relationship (SAR) data flows in real time. A medicinal chemist can review assay results the same day they are generated, adjust compound design immediately, and have the next iteration synthesised and tested within the same project cycle. This tight "design-make-test-analyse" (DMTA) cycle, is dramatically compressed compared to a multi-vendor model where data must be packaged, sent, received, interpreted, and acted upon across organisational boundaries.
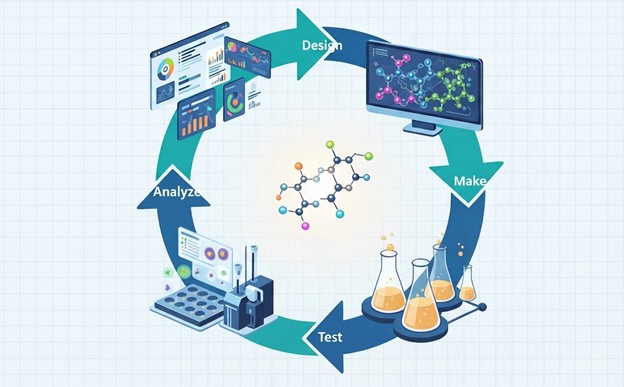
Organisations like Domainex, a Cambridge-based integrated drug discovery CRO, have demonstrated that a DMTA approach can save up to 30% on the average industry time from target to candidate molecule.
2. Reduced costs through operational efficiency
Managing a single CRO partnership eliminates the hidden costs of multi-vendor coordination: duplicate project management overhead, legal costs for multiple contracts, technology transfer fees, and the time your internal team spends acting as a data relay between providers. An integrated model consolidates these into a single, streamlined relationship.
3. Higher-quality scientific outcomes
In an integrated setting, multidisciplinary project meetings ensure that data from every discipline: enzymology, cellular biology, medicinal chemistry, computational chemistry, biophysics, DMPK, is considered holistically at every decision point. This prevents the common pitfall where a compound series is optimised for potency in isolation, only to fail later on selectivity, metabolic stability, or solubility grounds.
As Domainex notes, regular multidisciplinary project meetings ensure that new SAR data is shared, considered, and incorporated into the next iteration of compound design and testing, preventing information silos that can derail programmes.
4. Lower attrition risk
Attrition rates in drug development remain stubbornly high, with failure rates commonly cited at around 90%. A significant proportion of late-stage failures can be traced back to issues including poor pharmacokinetics, off-target toxicity or inadequate selectivity. An integrated approach is necessary to avoid such drawbacks before they become significantly problematic. By assessing ADMET properties, selectivity, and drug-like characteristics in parallel with potency optimisation from the earliest stages, an integrated CRO helps build quality into the pipeline from the outset.
5. Flexible engagement models
A strong integrated CRO offers flexibility in how you engage. You might begin with a discrete project focusing on fragment screening or assay development, before expanding into a fully integrated programme as the project matures. Alternatively, you might use a hybrid outsourcing model that blends your internal capabilities with the CROs specialist expertise, scaling up or down as the science demands.
What does the integrated drug discovery process look like at a CRO?
A typical integrated drug discovery programme progresses through five key phases, each building on the data and insights generated in the previous stage:
| Phase | Key Activities | Disciplines Involved |
|---|---|---|
| Target assessment & protein production | Construct design, protein expression and purification, Structural characterisation | Molecular biology, protein science, structural biology (X-ray crystallography, cryo-EM) |
| Hit identification | High-throughput screening (HTS), fragment screening, virtual screening, assay development | Assay biology, biophysics, computational chemistry, medicinal chemistry |
| Hit-to-lead | SAR exploration, selectivity profiling, early ADMET assessment, screening cascade design | Medicinal chemistry, assay biology, DMPK, computational chemistry |
| Lead optimisation | Multi-parameter optimisation of potency, selectivity, PK properties, safety profile | Medicinal chemistry, synthetic chemistry, DMPK, structural biology, cellular biology |
| Candidate selection | Final profiling, scale-up synthesis to support pre-clinical studies | Analytical chemistry, DMPK, synthetic chemistry, project management |
When you work with an integrated CRO, a dedicated project leader oversees the entire journey, ensuring continuity of knowledge and strategic direction. This single point of accountability is one of the most frequently cited advantages by organisations that have made the switch from multi-vendor to integrated outsourcing.
How do you choose the right integrated CRO partner?
Selecting an integrated CRO is a significant strategic decision. The following criteria should guide your evaluation:
- Breadth of in-house capabilities - Can the CRO genuinely deliver across biology, chemistry, structural biology, computational science, and DMPK, or will they sub-contract key elements?
- Depth of scientific expertise - Look for teams with significant industry experience (ideally 10+ years average per scientist) and a track record across multiple target classes and therapeutic areas.
- Track record of delivery - Ask for case studies, client testimonials, and evidence of programmes that have progressed to candidate nomination.
- Collaborative culture - The best CRO partnerships feel like an extension of your own team. Look for transparent communication, proactive problem-solving, and a willingness to challenge assumptions constructively.
- Client retention rates - High return rates are a strong proxy for client satisfaction. Organisations where over 80% of clients return for repeat engagements demonstrate consistent delivery.
- Flexibility - Can the CRO accommodate your preferred working model, whether that is a fully integrated programme, a hybrid model, or discrete service engagements?
Frequently asked questions about using a CRO for drug discovery
What is the difference between a CRO and a CDMO?
A CRO (contract research organisation) provides research and discovery services, from target identification through to candidate selection. A CDMO (contract development and manufacturing organisation) focuses on the development and manufacturing of drug substances and drug products, typically from the pre-clinical development stage onwards. An integrated drug discovery CRO operates in the early discovery space, upstream of CDMO activities.
Can a small biotech benefit from an integrated CRO?
Yes. Small biotechs and virtual pharma companies are among the primary beneficiaries of the integrated CRO model. Rather than building and maintaining expensive in-house discovery infrastructure, they can access world-class multidisciplinary expertise on a project basis, scaling their investment in line with programme milestones and funding rounds.
How does an integrated CRO handle intellectual property?
Reputable integrated CROs operate on a fee-for-service basis, with all intellectual property generated during the programme assigned to the client. This should be clearly established in the master services agreement before work begins.
What therapeutic areas can an integrated CRO support?
Leading integrated CROs work across a broad range of therapeutic areas, including oncology, CNS disorders, inflammatory diseases, cardiovascular disease, respiratory conditions, anti-infectives, and immuno-oncology. Their multidisciplinary teams also have experience across diverse target classes, including kinases, proteases, GPCRs, ion channels, protein-protein interactions, epigenetic targets, and emerging modalities such as targeted protein degradation and molecular glues.
How long does a typical integrated drug discovery programme take?
Timelines vary significantly depending on the target, the starting point, and the complexity of the programme. However, a well-executed integrated programme from hit identification through to candidate selection typically takes 2 - 4 years. The integrated model's primary advantage is compressing this timeline relative to what would be achievable through fragmented outsourcing.
What is a hybrid outsourcing model in drug discovery?
A hybrid outsourcing model combines a sponsor's internal capabilities with a CRO's specialist expertise and additional offshore support to move at speed. For example, a biotech with in-house biology might partner with a CRO for structural biology and medicinal chemistry while a third offshore partner, managed by the CRO, is responsible for accelerated synthetic chemistry, with all three teams operating as a single integrated project team. This model offers the benefits of integration while leveraging existing internal resources.
Why the integrated CRO model is the future of drug discovery outsourcing
The drug discovery landscape is evolving rapidly. Rising R&D costs, increasing scientific complexity and a growing pressure to deliver candidates faster are driving a structural shift away from fragmented, transactional outsourcing towards deeper, more strategic CRO partnerships.
Organisations that embrace the integrated model gain a compounding advantage: faster DMTA cycles, better informed decision making, stronger pipelines, and ultimately a higher probability of delivering candidates that succeed in clinical development. For biotech companies, pharmaceutical organisations, and academic groups navigating the complexities of early-stage drug discovery, partnering with an experienced integrated CRO is not just a tactical convenience, it is a strategic imperative.
Ready to explore how an integrated approach could accelerate your drug discovery programme? Contact Domainex to discuss your project with one of our experts.